FDA approves Yondelis in soft tissue sarcoma
The US FDA has approved Zeltia’s Yondelis (trabectedin) for the treatment of soft tissue sarcomas (STS) liposarcoma and leiomyosarcoma. This is positive news for Zeltia (MC:ZEL) and closely follows the approval of Yondelis in all types of STS in Japan late last month. Yondelis will be marketed in the US by Janssen (N:JNJ) and in Japan by Taiho (T:6470), and the approvals will trigger approx. $20m in milestone payments. The approvals are in line with our expectations and justify the recent strong share price, which is likely to continue to be buoyed by this positive news.
The FDA approved Yondelis as a second-line treatment of metastatic or unresectable liposarcoma and leiomyosarcoma for patients who have previously been treated with chemotherapy that contained anthracycline (eg doxorubicin). The approval is in line with our expectations, with no unexpected conditions.
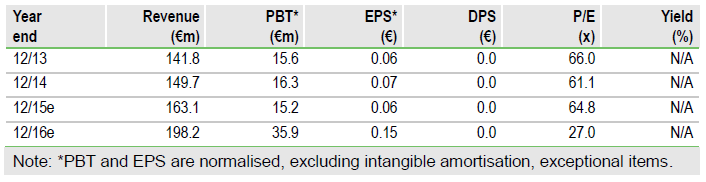
The Yondelis approvals will trigger milestone payments totalling approx. $20m ($10m US, $10m Japan). Approximately 12,000 cases of STS are diagnosed in the US each year and we forecast peak US sales of $130m for Yondelis in STS. Zeltia will receive a royalty on Yondelis sales in the US (we assume 15%) and a margin on sales of raw material to Janssen. The company’s trabectedin manufacturing facility in Madrid passed an FDA inspection in July and Janssen has purchased €5.5m of Yondelis active ingredient over the past two quarters, which could enable Yondelis to be launched in the US market before the end of November.
To Read the Entire Report Please Click on the pdf File Below